| CLICK HERE FOR INDEX PAGE | |
| OIL REFINERY | |
| V. Ryan © 2011 | |
| PDF FILE - CLICK HERE FOR PRINTABLE WORKSHEET | |
|
Crude oil is virtually unusable in its unrefined state. It is composed of a mix of hydrocarbons, each with a different number of carbon atoms. The hydrocarbons are different in weight, with the lightest being propane (gas) and heaviest asphalt (used on roads). Extracting the various hydrocarbons from crude oil is a formidable challenge and this process takes place in an oil refinery. The process is called ‘distillation’. |
|
 |
|
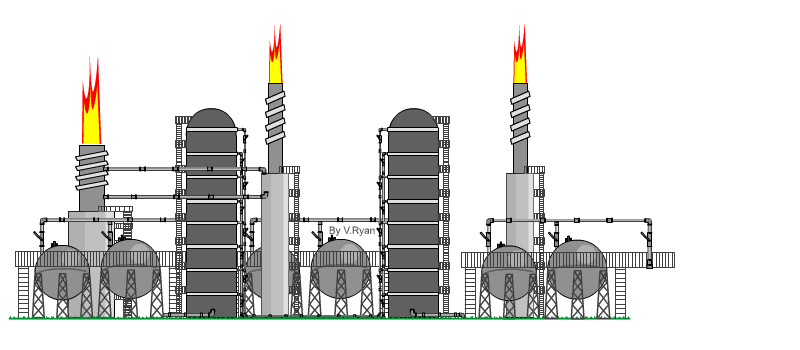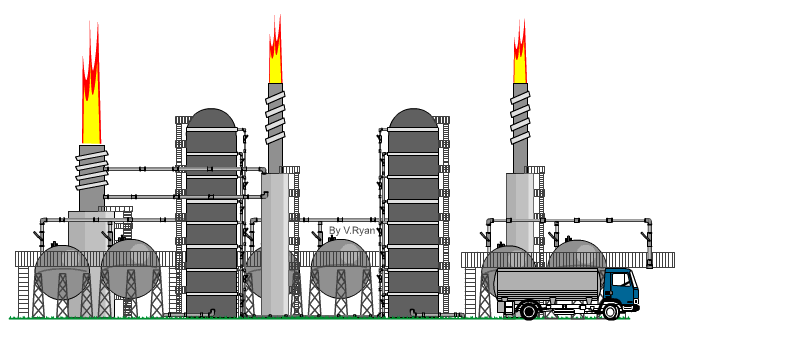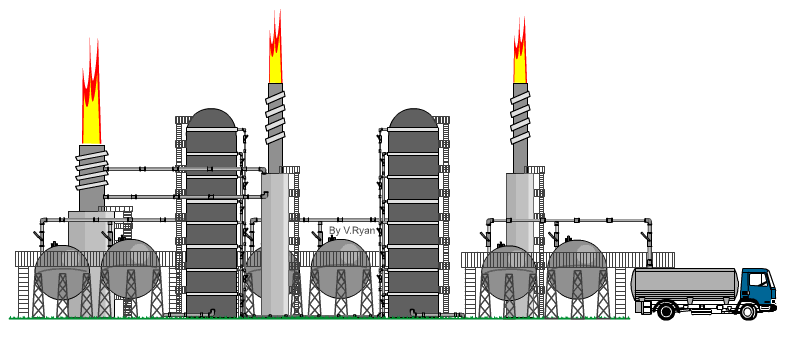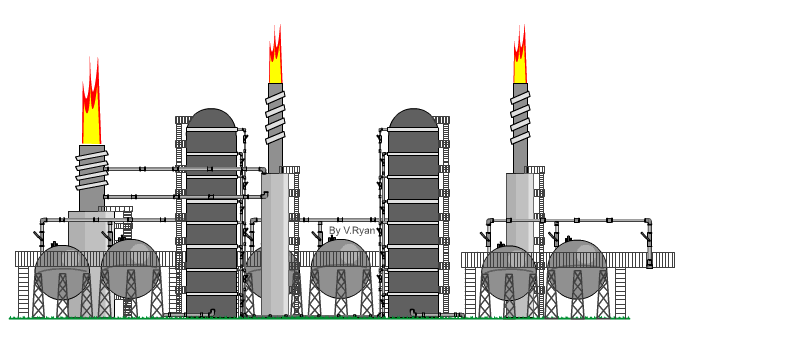
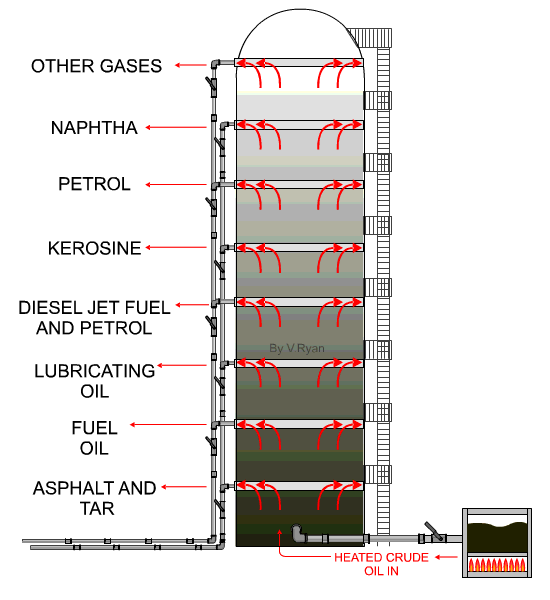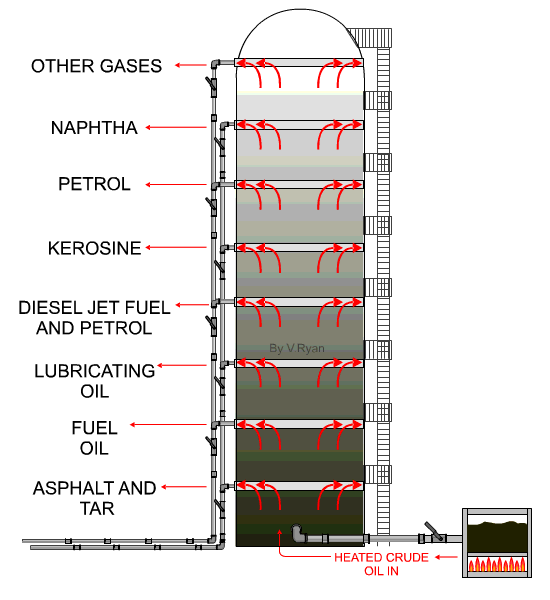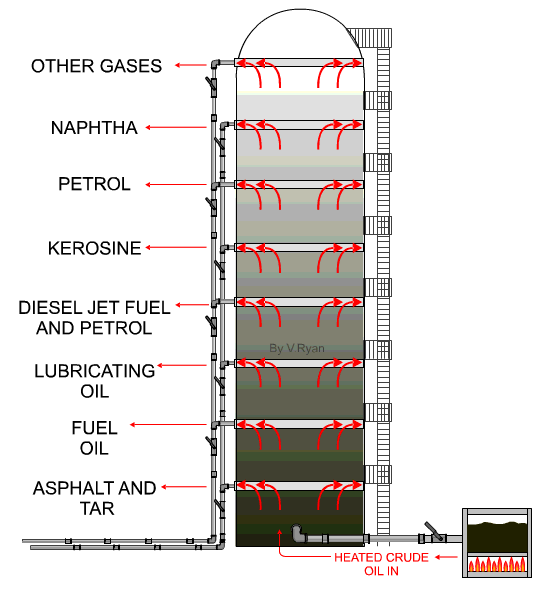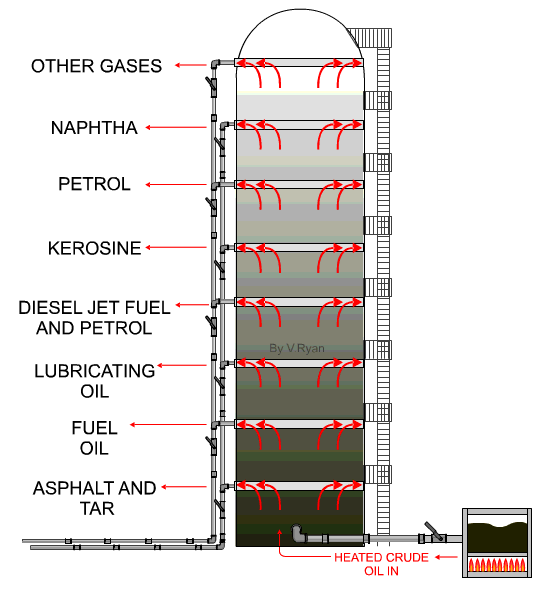
| The
distillation process takes place at an oil refinery in a distillation
‘Tower’. An example is seen opposite. When crude oil is refined it is ‘distilled’ into fractions. Each hydrocarbon extracted from crude oil, is called a fraction. The refining process is also called the ‘cracking’ process. Crude oil is heated to over 370 degrees Celsius and tit is pumped into the base of the distillation tower. The crude oil vaporises and rises up the tower. As it cools the molecules condense, with heavier molecules of asphalt, bitumen and tar at the bottom and lighter molecules such as diesel, petrol and aviation fuel at the top. At each level, the condensed hydrocarbons are siphoned off. The fractions / hydrocarbons produced include gas, petrol, diesel, heating oils, naphtha (important for plastics), ethylene and polyethylene. Bitumen / asphalt is also produced, as a residue. |
 |
 |
|
| CLICK HERE FOR RESISTANT MATERIALS INDEX PAGE | |