What is Anodizing? Anodizing is an electrochemical process, that converts the aluminium surface into a decorative, durable, corrosion-resistant, anodic oxide finish. Anodising also increases the wear resistant of the metal and produces a much better surface for the application of primers and paints. Anodising is often followed by dying, to produce an attractive, colourful finish. Anodised layers are non-conductive and provide improved adhesion for other finishes. An anodised surface is entirely integrated with the surface of the metal, it will not chip or peal away.
Anodising is a process through which a metal such as aluminium, receives a corrosion resistant, oxide finish (anodic finish). This is an oxide layer (aluminium oxide) created through electrolysis.
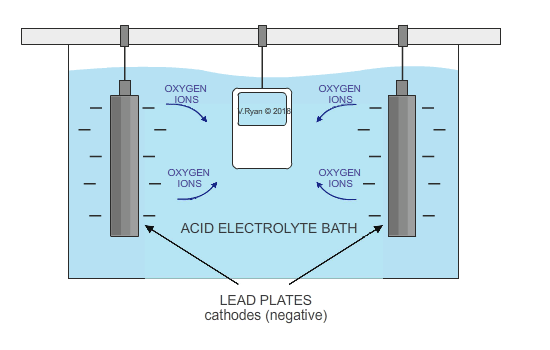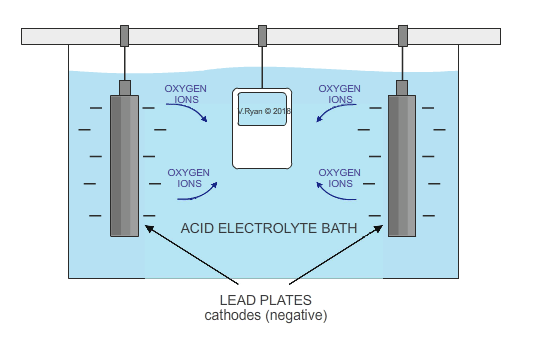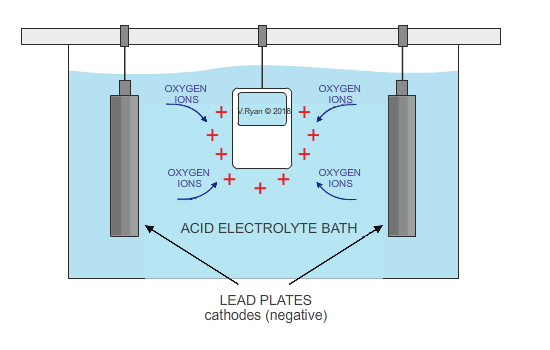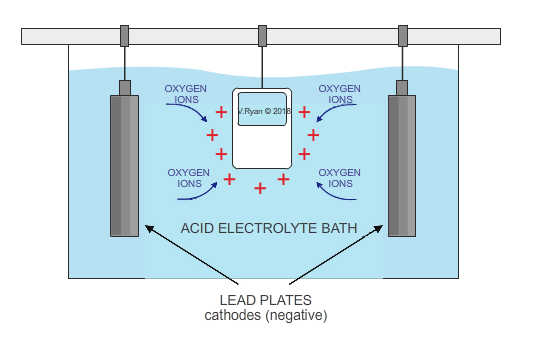
The metal, aluminium in this case, acts as the anode (positive electrode), as it is positively charged. It is submerged in a solution of sulphuric acid, sodium sulphate and water. Lead plates act as the cathode (negative electrode) and when electricity is allowed to flow, oxygen ions are released from the electrolyte. They combine with aluminium atoms at the surface of the anode, creating a thin oxide layer.