
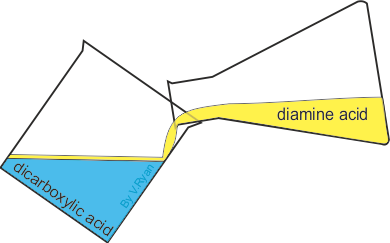
Equal amounts of diamine acid and dicarboxylic acid are carefully poured together.
The oil based diamine acid floats on top of the water based dicarboxylic acid.
| CLICK HERE FOR INDEX PAGE | ||
| WHAT IS NYLON AND HOW IS IT MANUFACTURED? | ||
| V.Ryan © 2015-16 | ||
| PDF FILE - CLICK HERE FOR PRINTABLE WORKSHEET | ||
| VIDEO - INTRODUCTION TO NYLON | ||
 |
||
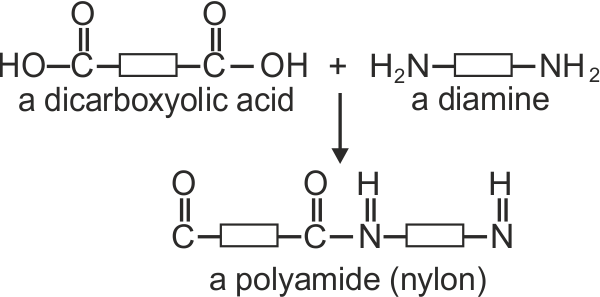
| Nylons are members of the family of polymers called ‘condensation copolymers’. These are long molecule polymers, that are formed through a condensation reaction (sometimes called ‘dehydration synthesis’). | ||
 |
||
| HOW NYLON IS MANUFACTURED IN A CLASSROOM | ||
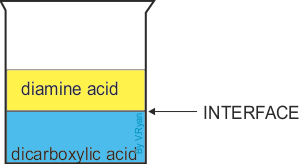
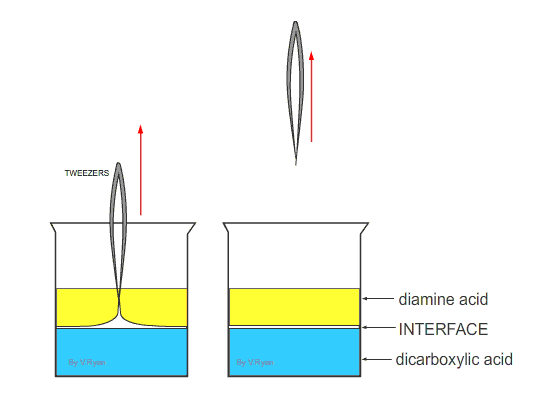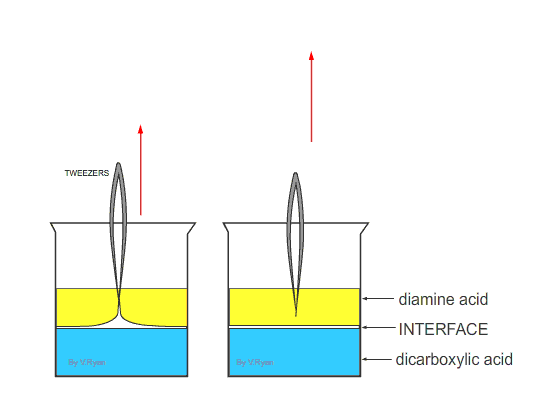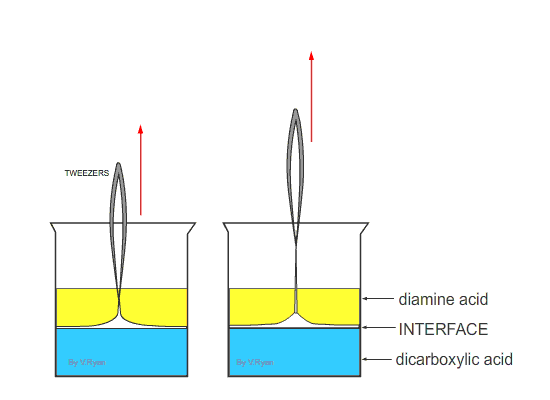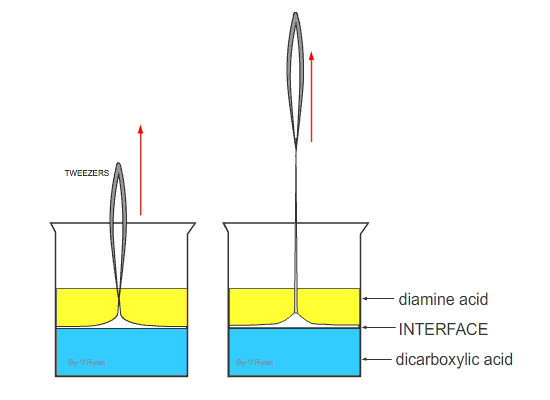
| Nylon fibre is produced when equal volumes of a diamine acid and a dicarboxylic acid are carefully and slowly poured together. One is oil based (diamine) and floats on the top of the other, water based chemical dicarboxylic. The molecules of both chemicals can bond together, forming long chains. A delicate film forms where the two chemicals meet (called the ‘interface’). This is called a ‘condensation reaction’. The film can be removed using tweezers and as it is removed a new boundary between the chemicals forms and process continues, forming long chains of nylon. | ||
 |
Equal amounts of diamine acid and dicarboxylic acid are carefully poured together. The oil based diamine acid floats on top of the water based dicarboxylic acid. |
|
 |
The interface forms where the acids meets (a condensation reaction) | |
| Tweezers are used to remove the film of polyamide (nylon). The polyamide will continue to form as long as the tweezers lift the film and as long as the two chemicals last. | ||
 |
||
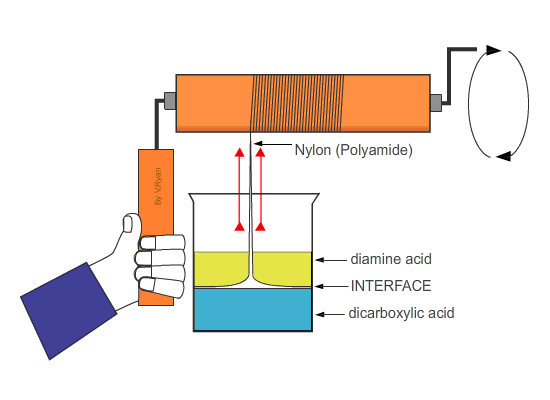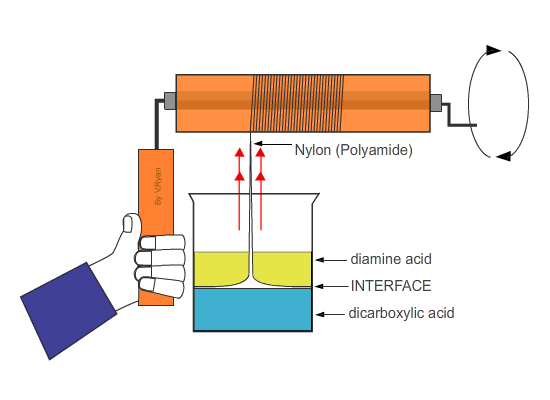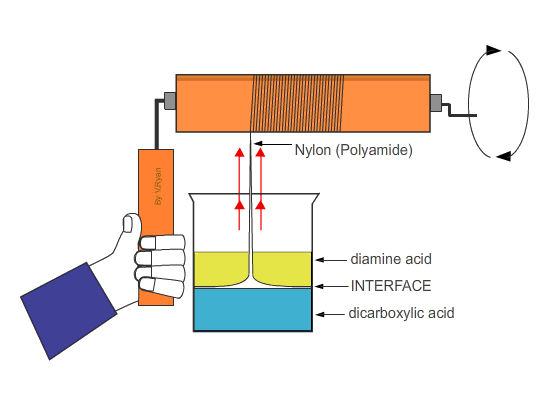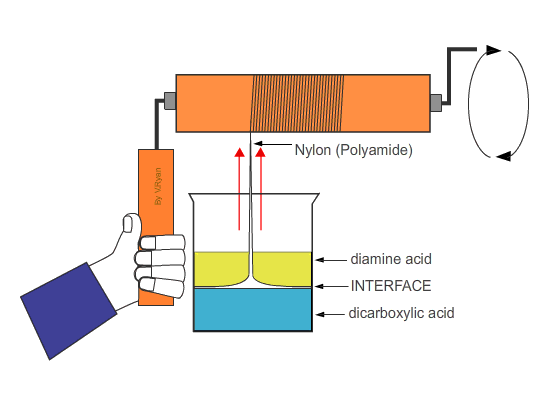
| If the nylon ‘thread’ is wrapped around a roller which is then rotated, long lengths can be produced. | ||
 |
||
| CLICK HERE FOR FIRST PAGE ON NYLON | ||
| CLICK HERE FOR RESISTANT MATERIALS INDEX PAGE | ||
|
||